Test labs
Test Labs is a dedicated test facility located at Tonsley within the College of Science & Engineering, Flinders University. The facility was established through funding received from Flinders’ Foundation and the South Australian Government through the Research Commercialisation and Startup Fund. The facility provides research and testing of Personal Protective Equipment (PPE) including surgical masks, respirators and medical gowns.
Australia-ASEAN Virtual Seminar Series: Personal Protective Equipment for COVID-19
The 'ASEAN Virtual Seminar Series: Personal Protective Equipment (PPE) for COVID-19' received grant funding from the Australian Government through the Australia-ASEAN Council of the Department of Foreign Affairs and Trade.
In this series of online seminars, delivered in 2021, a panel of experts from Australia and ASEAN member countries shared technical knowledge and experiences related to face mask and gown testing and manufacturing with universities and industry.
You can view the recordings of the seminars here:
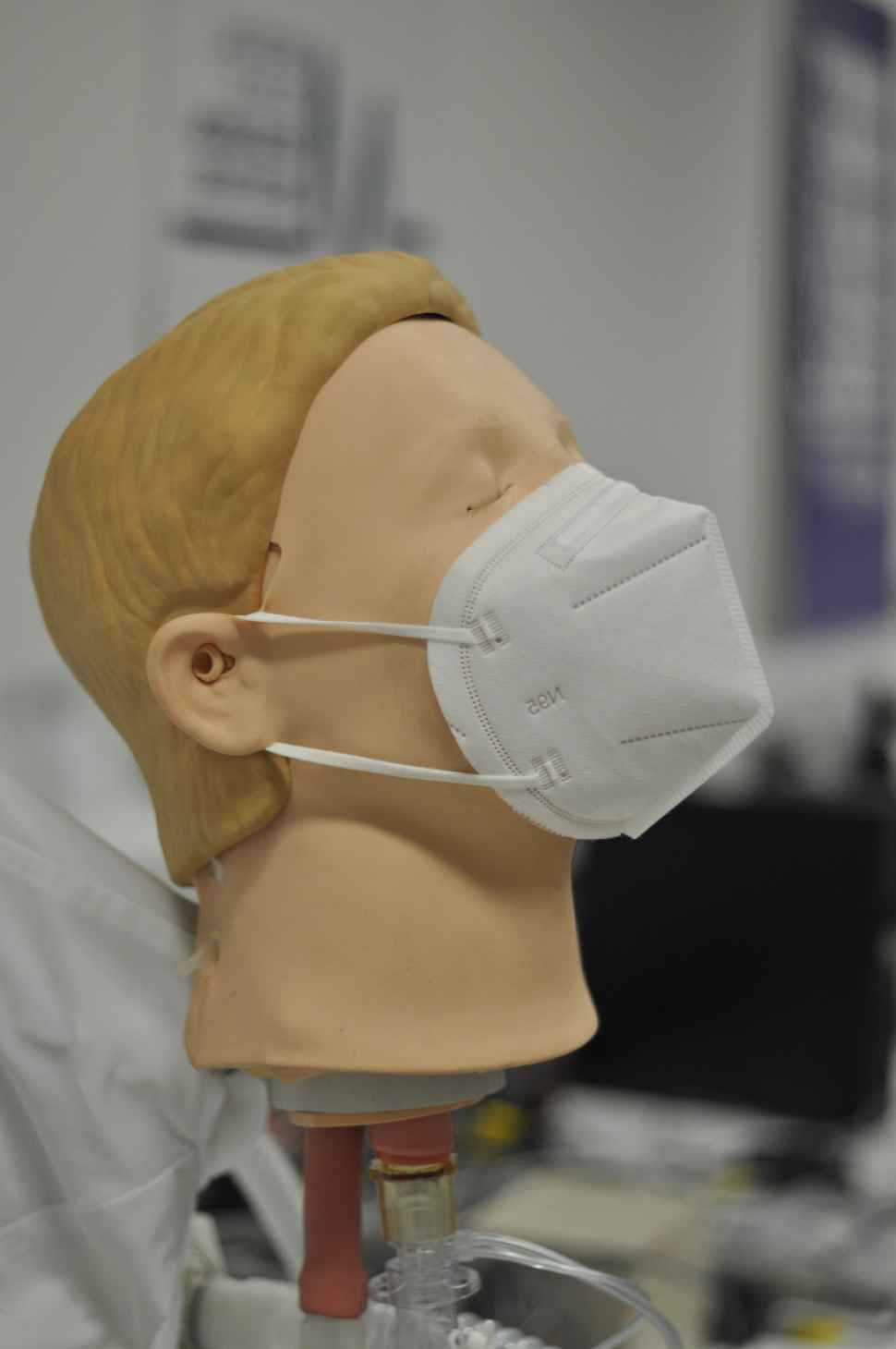
Image: Discussing correct face mask fitting in the PPE testing lab at Flinders University Tonsley campus (Photo Credit: Aaron Mohtar)
Standards for PPE
The following Standards apply to testing the performance of personal protective equipment within Australia and New Zealand:
Surgical masks and respirators
AS/NZS 4381:2015 ‘Single use face masks for use in health care’
U.S. National Institute of Occupational Safety and Health (NIOSH) 42 CFR 84 Respiratory Protective Devices
AS NZS 1716: 2012 Respiratory protective devices
ISO 22609: 2004 Clothing for protection against infectious agents – Medical face masks – Test method for resistance against penetration by synthetic blood
Surgical gowns
ANSI/AAMI PB70:2003 Liquid barrier performance and classification of protective apparel and drapes intended for use in health care facilities
ASTM 1670 Method for resistance of materials used in protective clothing to penetration by synthetic blood
ASTM 1671 Resistance of materials used in protective clothing to penetration by blood-borne pathogens using Phi-X174 Bacteriophage penetration as a test system
FAQs
A surgical mask is a fluid-resistant, single-use face mask that is worn over the nose, mouth and chin. These masks are intended for use in surgical, medical and dental procedures and are used to minimise exposure to others from infectious airborne organisms generated by the wearer. These masks are graded as Level I, II or II based on the level of protection they offer and their application.
Disposable filtering facepiece respirators or particle filtration respirators are filtering facepieces designed to reduce the inhalation exposure to particulate contaminants. In the health care environment, these masks are used to protect healthcare workers from infectious aerosolised droplets released from infected patients.
N95 and P2 refer to a class of filtering face piece or particulate respirator that has been certified according to the relevant Standard. AS/NZS 1716:2012 is the Australian / New Zealand Standard used to test the performance of a particle filtration respirator. The term P2 is applied to masks that are compliant with the Australian Standard. The National Institute for Occupational Safety and Health (NIOSH) in the United States of America uses the Standard 42 CFR 84, and classes these masks types as N95, N99 or N100, depending on their filtration efficiency.
Surgical gowns are single use personal protective garments intended to be worn by health care personnel during surgical procedures to protect the wearer from the transfer of microorganisms, body fluids and particulate matter.
Isolation gowns are single use personal protective garments intended to be worn to protect either patients or health care personnel and visitors from the transfer of infectious agents. They are required to have long sleeves and cuffs that cover the wearer to the wrist.
Sturt Rd, Bedford Park
South Australia 5042
South Australia | Northern Territory
Global | Online
CRICOS Provider: 00114A TEQSA Provider ID: PRV12097 TEQSA category: Australian University








