Infectious - TTANGO2 POCT Program
Test Treat ANd GO 2
Many remote Aboriginal and Torres Strait Islander communities in Australia experience unacceptable high rates of STIs such as chlamydia, gonorrhoea and trichomonas, despite efforts over many years to bring these infections under control.
A key public health strategy for control of curable STIs is the provision of accurate testing and timely treatment through primary care services.
Recent technological advances have led to the development of highly accurate point-of-care (POC) tests for STIs using the GeneXpert device, based on nucleic acid amplification technology (NAAT).
The TTANGO (Test Treat ANd GO) research trial was carried out from 2011-2016 to determine the acceptability, performance and short-term health impact of this new POC technology for the detection of chlamydia (CT) and gonorrhoea (NG) in remote communities. The TTANGO Trial was funded by an NHMRC Grant, for more information, visit the Kirby Institute website or view the TTANGO Trial Findings Report on the TTANGO2 website.
Building on the TTANGO Trial, TTANGO2 is a translational research program which is focused on the wider implementation of STI POC testing in high prevalence areas in regional and remote Australia
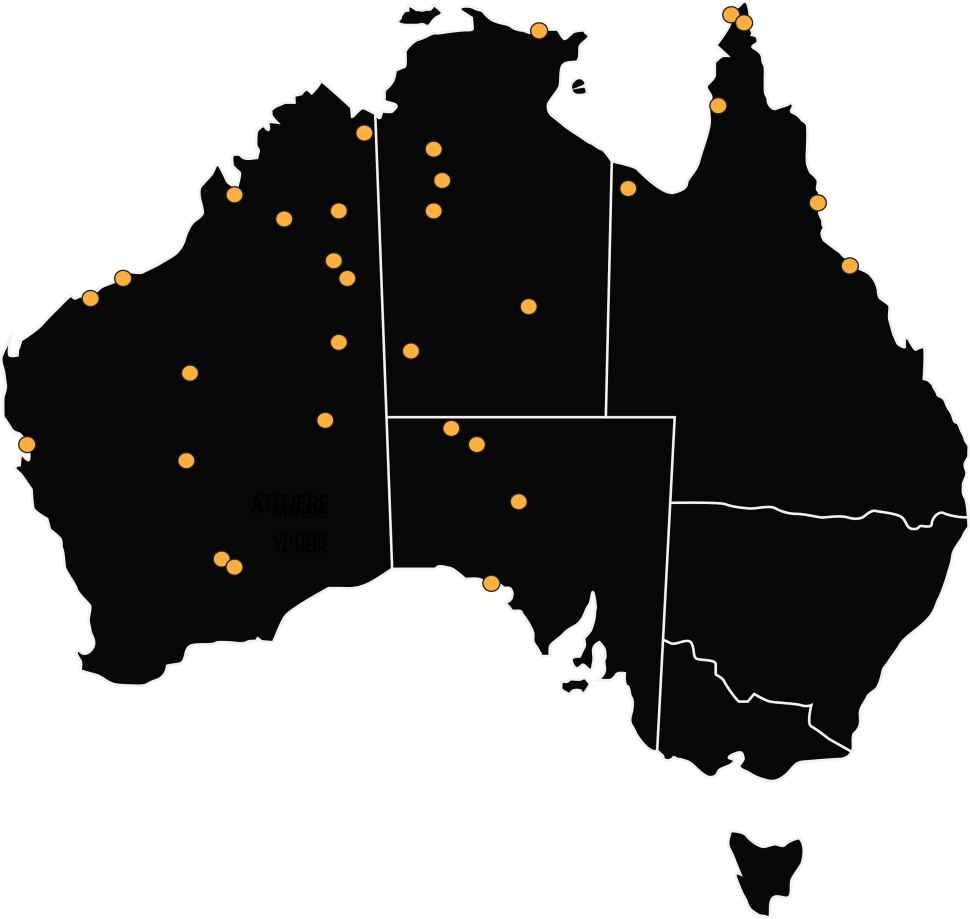
Through TTANGO2, STI POC testing for chlamydia, gonorrhoea and now trichomonas on the Cepheid GeneXpert is being undertaken in a network of 32 primary health services over a 5-year period to evaluate the long-term uptake, sustainability and impact of STI POC testing in these services.
POC testing, as well as enhanced continuous quality improvement practices, has led to earlier detection and treatment uptake for many Aboriginal and Torres Strait Islander people.
TTANGO2 is a collaboration between the Kirby Institute, University of New South Wales, Flinders International Centre for Point-of-Care Testing, other academic research institutions, Aboriginal and government health organisations, pathology providers, health services, communities and industry. It is funded by an NHMRC Partnership Grant and the Australian Government Department of Health, WA Health and QLD Health.
Sturt Rd, Bedford Park
South Australia 5042
South Australia | Northern Territory
Global | Online
CRICOS Provider: 00114A TEQSA Provider ID: PRV12097 TEQSA category: Australian University